Clean Air Technology: Pharmacy Isolators, Pharmacy Cleanrooms & Fixtures, Chemo Hoods, Sterile Room Medication Preparation, Barrier Isolators, Chemo Gloveboxes...












Modular Cleanrooms knows cleanrooms can be constructed out of many varying materials from sheet rock to flexible vinyl, but no matter what material it is, the HEPA filter is the heart of every Cleanroom. That is why Modular Cleanrooms is a Master Distributor of Envirco Corp. and has the largest inventory in the central US of Mac 10 Fan Powered HEPA Filters. Along with the Mac 10 filters we stock several styles and sizes of cleanroom light fixtures (T8 with electronic ballasts) in standard Troffer and Flo-Thru styles, Suspended T Bar ceiling grid and ceiling tiles. We also stock replacement motors, pre-filters and HEPA filters (standard and RSR) for the Mac 10 series of Fan Filter Units (FFUs). Modular Cleanrooms is also able to purchase replacement HEPA filters directly from the manufacturers for most popular brands of FFUs.
The Econo-Tent softwall cleanroom is the most economical approach and design for your cleanroom needs. This design is commonly used in the microelectronics and medical mold injection industry as well as many others. Econo-Tent's softwall design allows it to be used as either a totally freestanding room or it can be used in combination with your existing walls and ceiling. Each room comes completely factory prefabricated for minimal on-site erection time and is totally self-contained with all components factory-finished.
Modular Hardwall Cleanrooms are prefabricated, factory engineered and designed to meet a full range of cleanroom requirements found in industries ranging from Micro-Electronics, Aerospace, Medical equipment and packaging. Modular Hardwall Cleanrooms are also an excellent product for the Compounding Pharmacy market. They meet and exceed all of the requirements found in USP 797.
The Aluminum Bio-Clean design emphasizes particle infiltration as well microbial contamination protection. The Aluminum Bio-Clean can be designed and certified to as clean as Class 10 (ISO 4) up to Class 100,000 (ISO 8) as well as Class 10,000 for USP 797. Each Aluminum Bio-Clean cleanroom can also be outfitted with Class 100 (ISO 5) work zones eliminating the need for (ISO 5) Clean Benches/Hoods.
The Micro-Clean cleanroom is specifically designed to meet requirements of the medical and pharmaceutical industries. Micro-Clean's design allows it to be used as either a totally freestanding room or it can be used in combination with your existing walls and ceiling. The Micro-Clean comes completely factory prefabricated for minimal on-site erection time and is totally self-contained with all components factory finished.
Visit our website for more information







For over 40 years, NuAire has consistently led the pharmacy compounding industry in safety standards and customer satisfac¬tion. We obsess over our customers needs and tailor unique solutions to meet those needs. With invaluable input from customers like you, we now offer a wide range of laminar airflow workstations (LAFWs), containment ventilated enclosures (CVEs), biological safety cabinets (BSCs), ultralow temperature (ULT) freez¬ers, Restricted Access Barrier Systems (RABS), and much more. With our cutting-edge robotic sheet metal facility, we can deliver you custom, innovative pharmacy compounding solutions in addition to the standard products that we sell.
The LabGard Class II Type A2 & Type B2 Biological Safety Cabinets (BSCs) effectively operate as Containment-Primary Engineering Controls (C-PECs) for sterile hazardous pharmacy drug compounding. C-PECs provide USP 800 compliant product, personnel, and environmental protection that can be externally exhausted after passing through HEPA filtration.
The PharmaGard Restricted Access Barrier System (RABS) series offers a physical barrier between personnel and product. The positive pressure Compounding Aseptic Isolator (CAI) protects the non-hazardous compounded sterile preparation inside, while our negative pressure Compounding Aseptic Containment Isolators (CACIs) offer containment for hazardous sterile preparations and a choice of a model that recirculates most air to the work zone or another that is total exhaust. NuAire offers accessories so that either of these CACI models can be connected to external exhaust in compliance with USP 800.
The AireGard Laminar Airflow Workstation (LAFW) series operates as a Primary Engineering Control (PEC) that complies with USP 797 by providing product protection by flushing the work surface with vertical or horizontal ISO class 5 air.
The LabGard NU-813 Containment Ventilated Enclosure (CVE) or Class I BSC also effectively operates as a C-PEC that complies with USP 800 for non-sterile hazardous drug compounding. The NU-813 offers personnel and environmental protection by creating a constant 80 fpm (0.41 m/s) air inflow that is exhausted through HEPA filters.
The Blizzard -86°C Ultralow Temperature (ULT) Freezer series provides consistent storage temperatures of -40°C to -86°C, ideal for vaccine storage, and are equipped with energy efficient compressor motors. All NuAire freezer models utilize environmentally friendly hydrocarbon (HC) refrigerants and vacuum insulated polyurethane (VIP) chamber walls. Available capacities range from 3.5 to 14.8 ft3 (100 to 828 L).
NuAire pharmacy compounding workstations can be fitted with an optional IV bar with 6+ SST hooks, mechanical auto-rising base stands, ergonomic arm attachments for computer monitor + keyboard/mouse, and glass sidewalls for added visibility.
• View the NuAire Platinum Pages Publication Ad








Baker has a long history of providing the highest quality containment and clean air products for retail, hospital and clinical pharmacy applications. Our products continue to set the industry standard for safety, performance, operating efficiencies and ergonomics through innovative research, meticulous engineering and on-going testing.
With pharmacy laws, guidelines and regulations targeting the preparation of sterile injectable drugs, clean air and containment equipment is playing an increasingly critical role in day-to-day pharmacy operations. Laminar-flow clean benches, gloveboxes and biological safety cabinets are required in-house elements for minimizing potential contamination and exposure during compounding.
For 60 years The Baker Company has been helping our customers advance science, discovery, and clinical care by providing the right equipment to meet their needs. We look forward to working with you!
We are committed to helping you protect your patients and employees, and offer a full range of products to help you be compliant with USP <795>, <797>, and <800> including:
PHARMACY GLOVE BOXES
Positive pressure sterility assurance barrier glove box for complete product protection when compounding non-hazardous pharmaceutical compounds and related clinical, pharmacy and process applications.
For sterile compounding of hazardous or potent pharmaceutical compounds, chemotherapy agents and IV admixtures that can be harmful to pharmacy personnel.
BIOSAFETY CABINETS
The most energy efficient, comfortable and safe A2 cabinet in the industry, the SterilGARD e3 is designed for sterile product preparation and biological experimentation involving agents of low to moderate risk. Also available is the compact model SG-303, a 3-foot biosafety cabinet with a smaller footprint for low-volume facilities.
The BioChemGARD® e3 is a breakthrough in total-exhaust biological safety cabinets. Engineered specifically for laboratories that need containment and removal of vapors, mists and particulates, the BioChemGARD e3 has a revolutionary airflow system that increases users comfort and productivity while decreasing the overall operating costs. Designed for biological testing and product preparation involving low to moderate risk agents where chemical effluent is present and clean air is essential.
LAMINAR FLOW CLEAN BENCHES
With a spacious work area and brightly illuminated for maximum productivity and unique high velocity air return slots, EdgeGARD® HF Clean Bench offers both user comfort and product protection. Horizontal and vertical flow models are available.
OPTIONAL REMOTE COMPOUNDING VALIDATION PACKAGE
Phocus Rx remote compounding validation system from Grifols is a plug & play solution that provides a safe, reliable and easy-to-use environment to document and validate the compounding of prescription IV drugs. The system improves quality assurance and workflow efficiency by helping to ensure that medications are prepared correctly and labels are accurate. Baker biosafety cabinets and clean benches are available fully integrated with Phocus Rx.
• Visit the The Baker Company Website







Esco Lifesciences is a world-leading life science company with a diversified portfolio and sales in over 100 countries. As a manufacturer of laboratory and biopharma equipment, and IVF medical devices, Esco offers tailored solutions that fit the needs of laboratories in various industries.
Esco Lifesciences contributes to meet the challenges of the 21st century with a diverse range of business units. The company continuously innovates its products to help the clinical and industrial laboratories achieve successful conclusions in research and development, quality control, and analysis. We continue to provide reliable world-class equipment to help pharmaceutical companies make their products safer and more cost-effective
Time is running out, but you can rely on Esco for a one-stop shop for equipment to comply to USP 800.
USP 800 is officially enforced by local, state, and federal regulatory agencies that require compounding pharmacies to comply, or otherwise face a hefty fine or even closure. Hazardous drugs must be compounded in a proper containment primary engineering control (C-PEC) such as biosafety cabinet (BSC) or compounding aseptic containment isolator (CACI) that is externally vented and located in an ISO Class 7 cleanroom.
• View the ESCO Platinum Pages Publication Ad






Modular Clean Rooms
Northwell Health Relies on Modular Devices Inc...
Veltek Associates
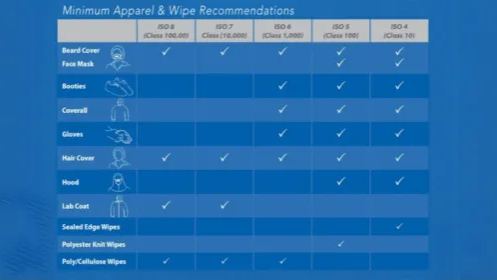
Environmental monitoring is a process that is used to monitor the microbial and particulate levels in a classified area...
NuAire
To ensure a successful HD facility design, first consider the pharmacy's business model, with a focus on compounding volume...
NuAire
Nonsterile hazardous drug handling processes
















































About Barrier Isolators (for Hospital Pharmacies and Hospital Labs)
From Wikipedia 2010
A barrier isolator, or simply an isolator, is a device that provides a physical barrier between a pharmacist or technician and a work process. Isolators are routinely found in the pharmaceutical industry within hospitals, and with the recent implementation of USP 797 ("Pharmaceutical Compounding: Sterile Preparations", a set of sterile compounding standards issued by the United States Pharmacopeia), are increasingly used in pharmacy applications. They are designed to provide an isolation of a process or the maintenance of an internal condition (e.g., sterile or aseptic). Isolators may operate at positive, negative, or ambient differential pressure. Isolators may provide personnel, product, or environmental protection, or any combination thereof. They are used throughout industry, from orange juice filling lines to cytotoxic drug compounding to electronics manufacturing.
Regarding pharmacy applications within hospitals, because people are the greatest source of contamination during aseptic manufacturing of drugs, reducing personnel interventions into the process zone has significant impact on the efficacy of the final drug product. In the mid 1980's, the industry began to employ barrier isolators, and later, in the 1990s, Restricted Access Barrier Systems, or RABS, to separate people from the process. The acronym RABS was coined by Stewart Davenport of Upjohn (now Pfizer). Since that time, the technology and applications of these systems has developed and broadened significantly.
Connect with thousands of pharmacy professionals throughout every practice setting.
The server is experiencing a problem at this time. Try again later.